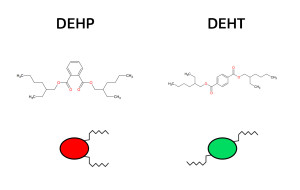
DEHT and DEHP isomer molecules have the same number of atoms: C24H38O4,but a different structure (structural formulae) and different physical and chemical properties. This shows how important structural differences are in determining a compound’s metabolism and toxicity. DEHP and DEHT are very different from each other in this respect.
DEHT is completely hydrolyzed: The ratio of 2-ethylhexanol to terephthalic acid is 2 mol to 1 (Barber et al.). Two other terephthalic esters: di-methyl-(DMT) and di-butyl-(DBT) undergo the same complete hydrolysis (Kamendulis et al.). It can therefore be concluded that complete metabolism takes place in the para position, while in ortho position in the case of DEHP, this does not happen.
This difference appears to be crucial in the case of the toxicological safety of medicinal products containing DEHP (in products containing PVC) and other products made for children. Phthalates enter the body in the large part orally, dermally and by inhalation. What’s more, compounds which enter the body by inhalation, exhibit much more severe toxic effects. So far, however, the role of phthalates in the formation of mental disorders has not been explained, nor has its mechanism of action in the nervous system.
Di(ethylhexyl) terephthalate (DEHT) is a general purpose plasticizer, while the structural isomer bis(2-ethylhexyl) phthalate (DEHP) is known for its reprotoxicity. EU Regulation No 143/2011 of 17.2.2011 classifies DEHP, BBP and DIBP DIB phthalates as very high risk substances subject to a permit issue procedure due to their harmful effects on reproduction (category 1B). However, it should be noted that the use of DEHP for the production of immediate packaging for medicinal products covered by Regulation (EC) No 726/2004, Directive 2001/82/EC or Directive 2001/83/EC (medical devices) does not require a permit.
Concerns about the potential risk of DEHP have led most companies to replace DEHP with DEHT. DEHT is its main replacement which is free from phthalates. It is widely available and tested, and has a comprehensive toxicological portfolio, which indicates that it can be used in toys, beverage packaging and medical devices (more on this topic can be found in subsequent articles).
It should also be mentioned that on 2 October of this year, the chemical company Grupa Azoty ZAK S.A. informed on their website that the phthalate-free Oxoviflex ® (DEHT) plasticizer meets the legal requirements of the European Union relating to the use of substances in products which come in contact with food and toys, thereby encouraging to become familiar with the test results on the substance. At the same time, the company is one of the most experienced manufacturers of plasticisers used in the processing of PVC in Europe. The phthalate-free and phthalate-based plasticizers they produce are sold under the names Oxoviflex ® and Oxoplast ®.